Centre for Biomedical AI
Harnessing AI to transform medical research and healthcare.

About the Centre
Artificial Intelligence (AI) is opening new frontiers in medicine, from diagnosing diseases to guiding personalised treatment decisions.
At the Centre for Biomedical AI, we’re using AI to design and evolve entirely new proteins — ‘designer’ proteins that could transform how we treat or even cure disease. By combining advanced algorithms with world‑class laboratory science, we’re creating next‑generation therapies with the potential to regenerate heart tissue after a heart attack, develop personalised cancer treatments, and engineer stem‑cell–based therapies for conditions like type 1 diabetes.
Centre Head
Laboratories
The laboratories operating within this centre are:
Hesselson Laboratory
Student opportunities
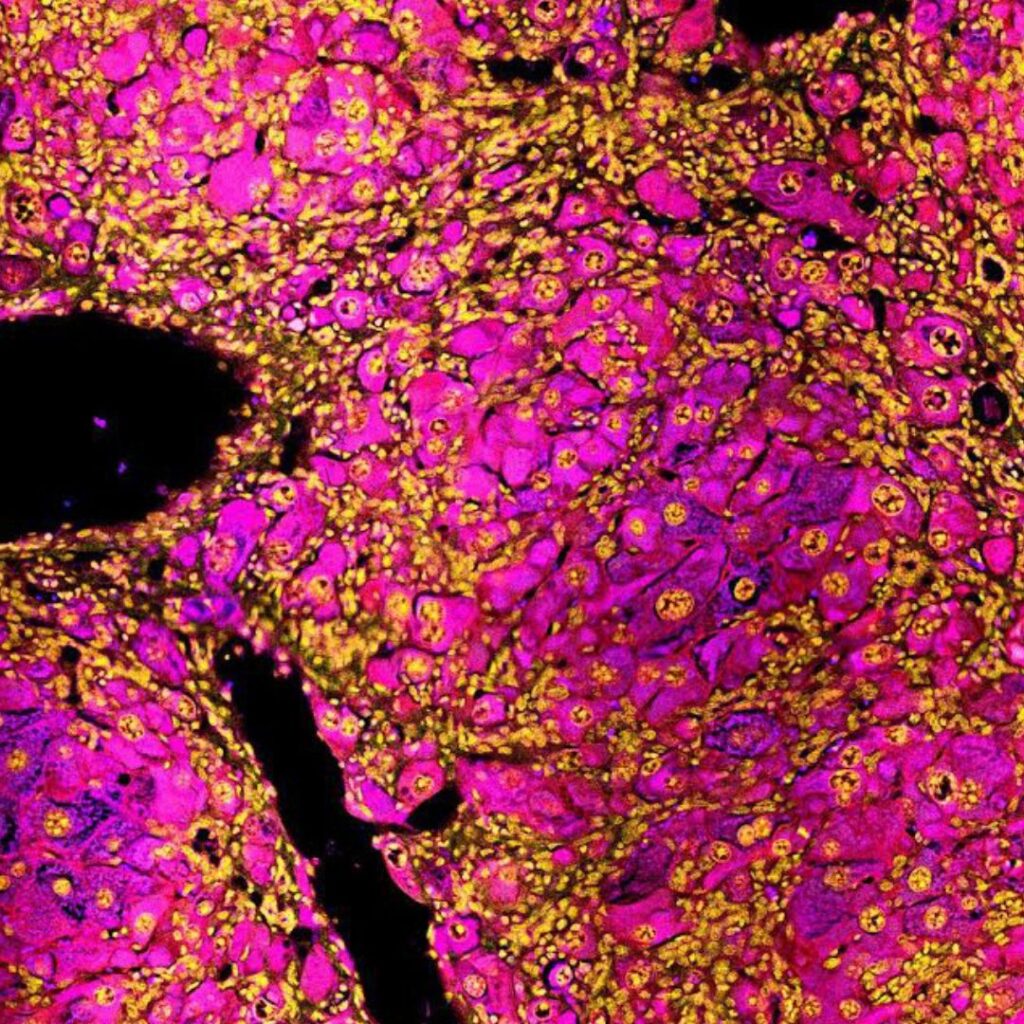
Directed evolution of liver hormones to enhance metabolism
Projects: Honours & PhD
Project details
Primary Supervisor: Associate Professor Daniel Hesselson
Supervisory Team: Dr Alex Cole
Centre: Biomedical AI
The hormone FGF21 is being developed as a therapy for type 2 diabetes and fatty liver with clinical trials showing promise for improving metabolic health including lowering liver fat and blood cholesterol. Natural FGF21 has a short-half life which has been extended by protein engineering to resist degradation or clearance from the body. However, stabilised FGF21 variants are typically less potent. This project will apply cutting edge directed evolution approaches to enhance the signaling activity (potency) of FGF21. The primary expected outcome is to produce a superior FGF21 molecule that could be commercialised as a fatty liver and/or diabetes therapy.
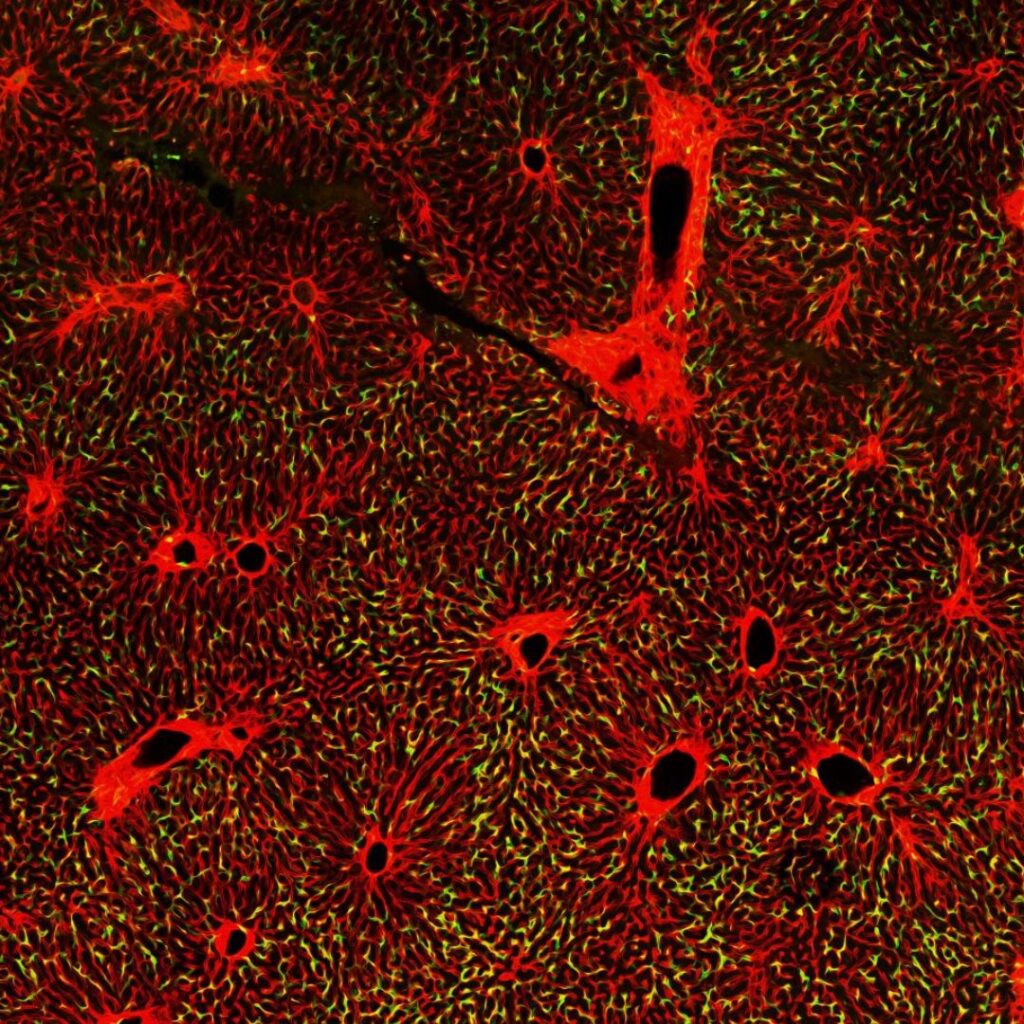
Enhancing the durability of stem cell-derived islets for type 1 diabetes
Projects: Honours & PhD
Project details
Primary Supervisor: Associate Professor Daniel Hesselson
Supervisory Team: Lise Hunault
Centre: Biomedical AI
Type 1 diabetes (T1D) results from the autoimmune destruction of insulin-producing beta cells, requiring continuous management of blood sugar levels. A lifelong cure for T1D will require regeneration or replacement of these cells. This project focuses on the transcription factor PDX1, which is a master regulator of pancreatic cell fate and is also essential for promoting optimal beta cell function. Excitingly, PDX1 both promotes regeneration and is critical for converting other pancreatic cell-types into beta cells. Our team uses directed evolution to engineer novel proteins in cells that model human disease. We have developed PDX1 variants with new properties that could enhance the function of stem cell-derived islets, which are the only renewable source of beta cells for curing T1D. The goal of this project is to determine which variant has the most potential for increasing the durability and function of transplanted islets, setting the stage for clinical translation.
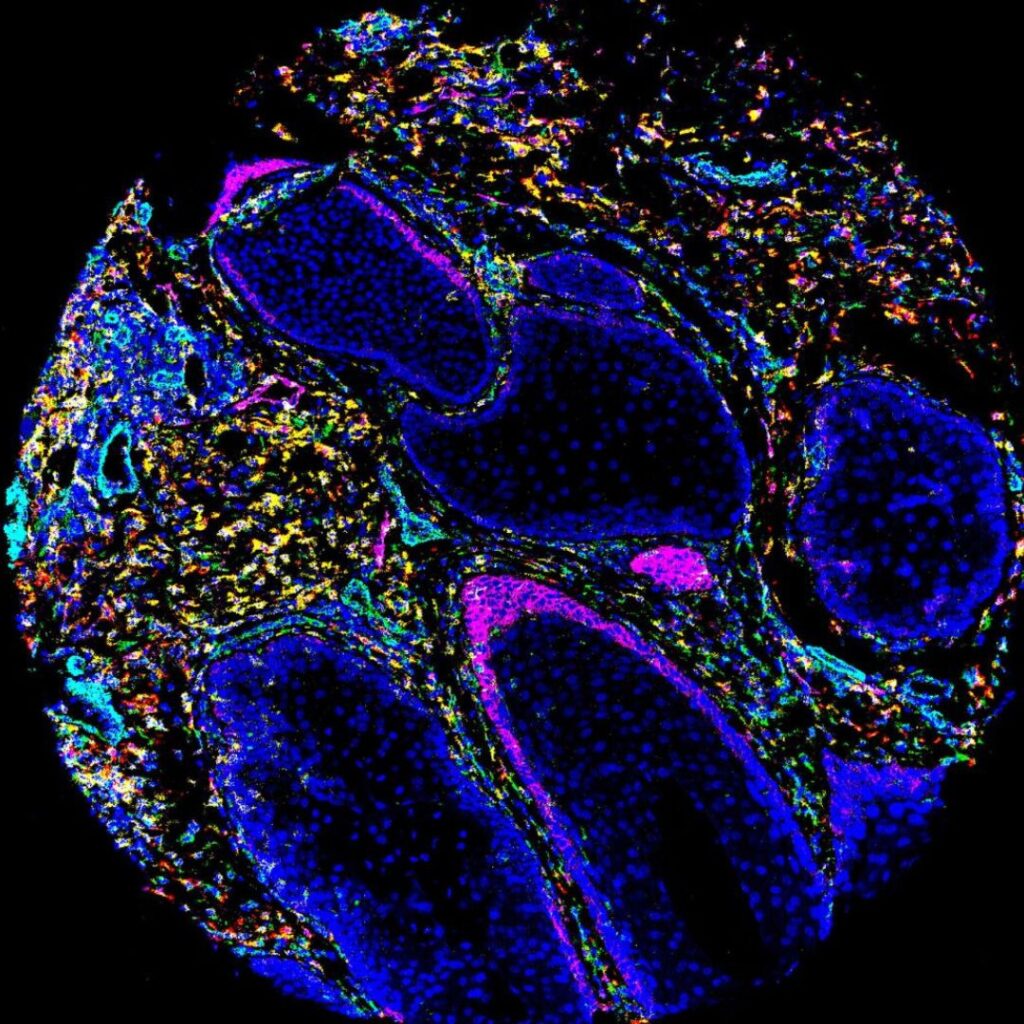
Tumour-activated cancer immunotherapies
Projects: PhD
Project details
Primary Supervisor: Associate Professor Daniel Hesselson
Supervisory Team: Dr Alex Cole
Centre: Biomedical AI
Solid tumours create an immunosuppressive microenvironment that limits the efficacy and safety of current immunotherapies, often leading to poor efficacy and off-target toxicity. This project aims to engineer next-generation protein and cellular therapies that are triggered by tumour-restricted signals such as hypoxia, acidic pH, or matrix metalloproteinase activity. Using our directed evolution and rational protein design platform, you will develop and test novel therapies in cell-based models, then evaluate them for safety and therapeutic benefit in mouse models of solid cancer. The goal of this research is to identify the most promising tumour-activated immunotherapeutics, laying the groundwork for more potent and less toxic cancer treatments.
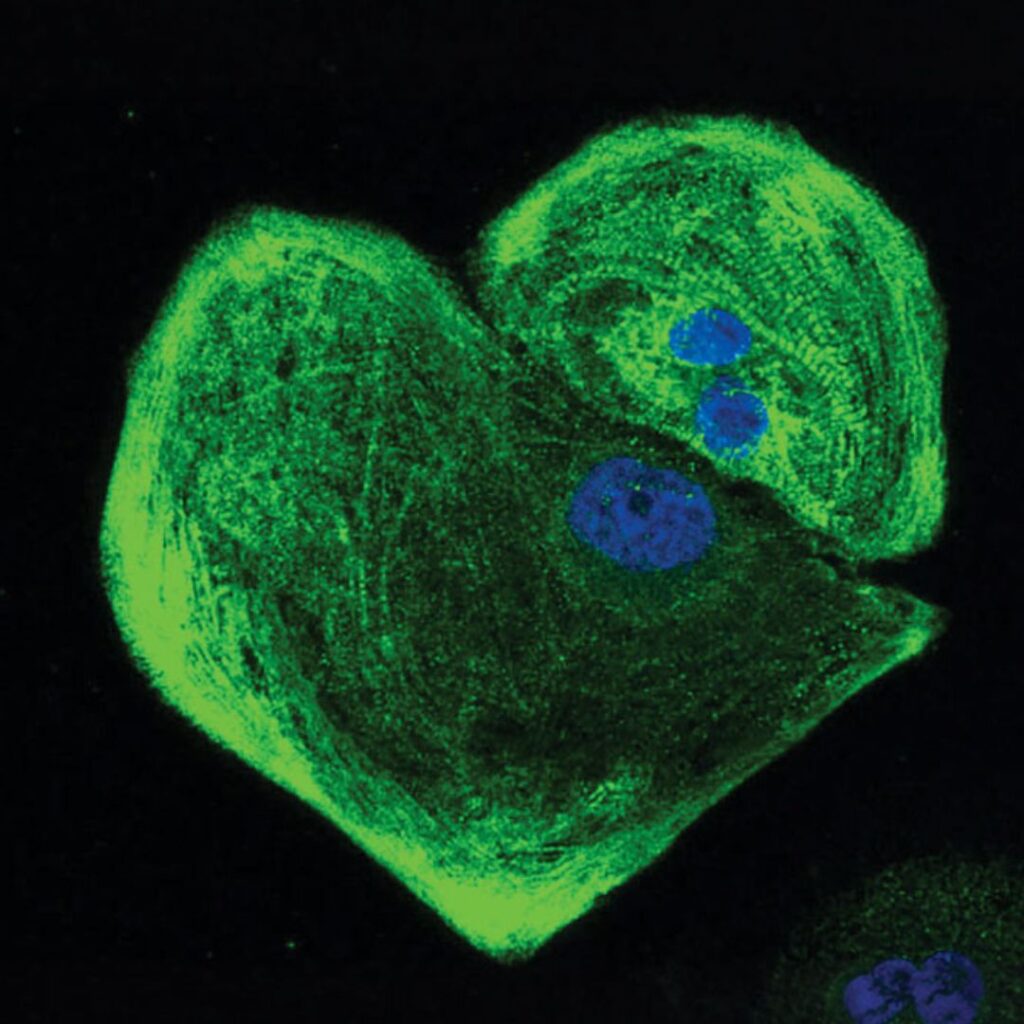
Novel delivery systems to empower complete repair after a heart attack
Projects: Honours & PhD
Project details
Primary Supervisor: Associate Professor Daniel Hesselson
Supervisory Team: Manuela Florido
Centre: Biomedical AI
Myocardial infarction (MI) results from the sudden loss of blood flow to cardiac tissue, causing massive cardiomyocyte death and subsequent scar formation that impairs pump function. Current clinical interventions stabilise patients and limit further damage but cannot regenerate lost myocardium. This project targets that unmet need by engineering protein therapeutics and delivering them at the optimal time and place to maximise cardiomyocyte survival, proliferation, and neovascularisation. Using our directed evolution and rational design platform, we have already generated lead protein candidates. The aim of this research is to deliver these candidates in in vitro human cardiomyocyte cultures and in vivo rodent MI models to identify the most potent regenerators, setting the stage for future preclinical and clinical translation.
Scholarship Opportunities
Biomedical AI PhD Awards
The purpose of these top-up scholarships is to support exceptional PhD students interested in applying cutting-edge computational and directed evolution approaches to produce novel therapeutics. Scholarships are available for PhD projects focused on cardiovascular disease or cancer. An annual top-up stipend of $20,000 (up to three years) will be offered to scholars who receive a Research Training Program (RTP, or equivalent) scholarship. For more information and to apply please contact biomedicalAI@
centenary.org.au
Get in touch
To get in touch and for all general enquiries relating to our work, please contact Associate Professor Daniel Hesseldon.


